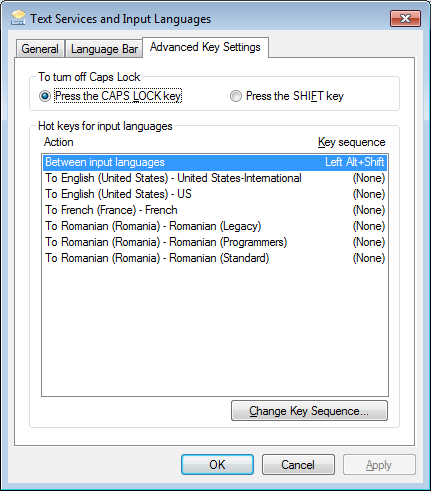
A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M .. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. A tenfold dilution for each step is called a logarithmic dilution or log-dilution, a 3.16-fold (100.5-fold) dilution is called a half-logarithmic dilution or half-log dilution, and a 1.78-fold (100.25-fold) dilution is called a quarter-logarithmic dilution or quarter-log dilution. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
In other words by doing two 1/5 dilutions. Our final dilution ends up being a 1/25 dilution. In other words our sample has been diluted 25 fold. = 25) This means that in this, there is 1 volume part sample to 24 volume parts of water for a total of 25 parts. If we wanted to work backwards. If our diluted sample was 0.12 M and it had been. 2-fold dilution is a bit confusing, a better way of describing dilution is ratio. For example, 2 fold dilution equals to 1:2 dilution. Since you mentioned serial dilution, you do this by first mixing an equal volume of bacteria and water (whatever used for dilution), and then mix an equal volume of the first mixture and water. A very quick lesson on how to do the math for serial and simple dilutions. Most Probable Number (MPN) Method - Duration: 7:05. Dilution methods and Making a table in P3.
In biology and medicine[edit]
In biology and medicine, besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. As, for instance, the number and size of bacterial colonies that grow on an agar plate in a given time is concentration-dependent, and since many other diagnostic techniques involve physically counting the number of micro-organisms or cells on specials printed with grids (for comparing concentrations of two organisms or cell types in the sample) or wells of a given volume (for absolute concentrations), dilution can be useful for getting more manageable results.[1] Serial dilution is also a cheaper and simpler method for preparing cultures from a single cell than optical tweezers and micromanipulators.[2]
In homeopathy[edit]
Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain.[3][4]
See also[edit]
References[edit]
- ^K. R. Aneja. Experiments in Microbiology, Plant Pathology and Biotechnology. New Age Publishers, 2005, p. 69. ISBN81-224-1494-X
- ^Booth, C.; et al. (2006). Extremophiles. Methods in microbiology 35. Academic Press. p. 543. ISBN978-0-12-521536-7.
- ^Weissmann, Gerald (2006). 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. The FASEB Journal. 20 (11): 1755–1758. doi:10.1096/fj.06-0901ufm. PMID16940145. Retrieved 2008-02-01.
- ^Ernst, Edzard (November 2005). 'Is homeopathy a clinically valuable approach?'. Trends in Pharmacological Sciences. 26 (11): 547–548. CiteSeerX10.1.1.385.5505. doi:10.1016/j.tips.2005.09.003. PMID16165225.
Two Fold Serial Dilution Method Test
- Michael L. Bishop, Edward P. Fody, Larry E. Schoeff. Clinical Chemistry: Principles, Procedures, Correlations. Lippincott Williams & Wilkins, 2004, p. 24. ISBN0-7817-4611-6.
4 Fold Serial Dilution
External links[edit]
- How to Make Simple Solutions and Dilutions, Bates College
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Serial_dilution&oldid=904833671'
A ten-fold dilution reduces the concentration of a solution or a suspension of virus by a factor of ten that is to one-tenth the original concentration. A series of ten-fold dilutions is described as ten-fold serial dilutions. In this manual, ten-fold serial dilutions are used in titrations of a suspension of Newcastle disease virus to establish the infectivity titre. They are carried out in small sterile test tubes. These tubes are usually made of glass and it is preferable if they have fitted lids to minimize the risk of contamination during the dilution.
A ten-fold dilution
Step 1. | Use a micropipette to dispense 900 mL of the diluent to glass tube. |
Step 2. | Use a micropipette to transfer 100 mL of the test solution to the first well. Discard the tip. |
Step 3. | Mix by shaking by hand or using a vortex mixer. |
Step 4. | Canoscan toolbox windows 10 download. The well now contains 100 mL of the original test solution diluted by one tenth in a total volume of 1000 mL. |
Ten-fold serial dilutions
Step 1. | Set up the sterilized glass test tubes in a rack. Label each tube clearly to indicate the dilution of its contents after the ten-fold serial dilution has been carried out. |
Step 2. | Use a micropipette to dispense 900 mL of the diluent to all the labeled sterile tubes. |
Step 3. | Use a micropipette to transfer 100 mL of the test solution to the first tube and mix. This is the first ten-fold dilution. |
Step 4. | Use a micropipette with new sterile tip to carry out a second ten-fold dilution. |
Step 5. | Continue the series of ten-fold dilutions until the last tube. |